InstantQMS helps manufacturers manage quality, meet regulations, and track
every step of their process in one easy-to-use system.
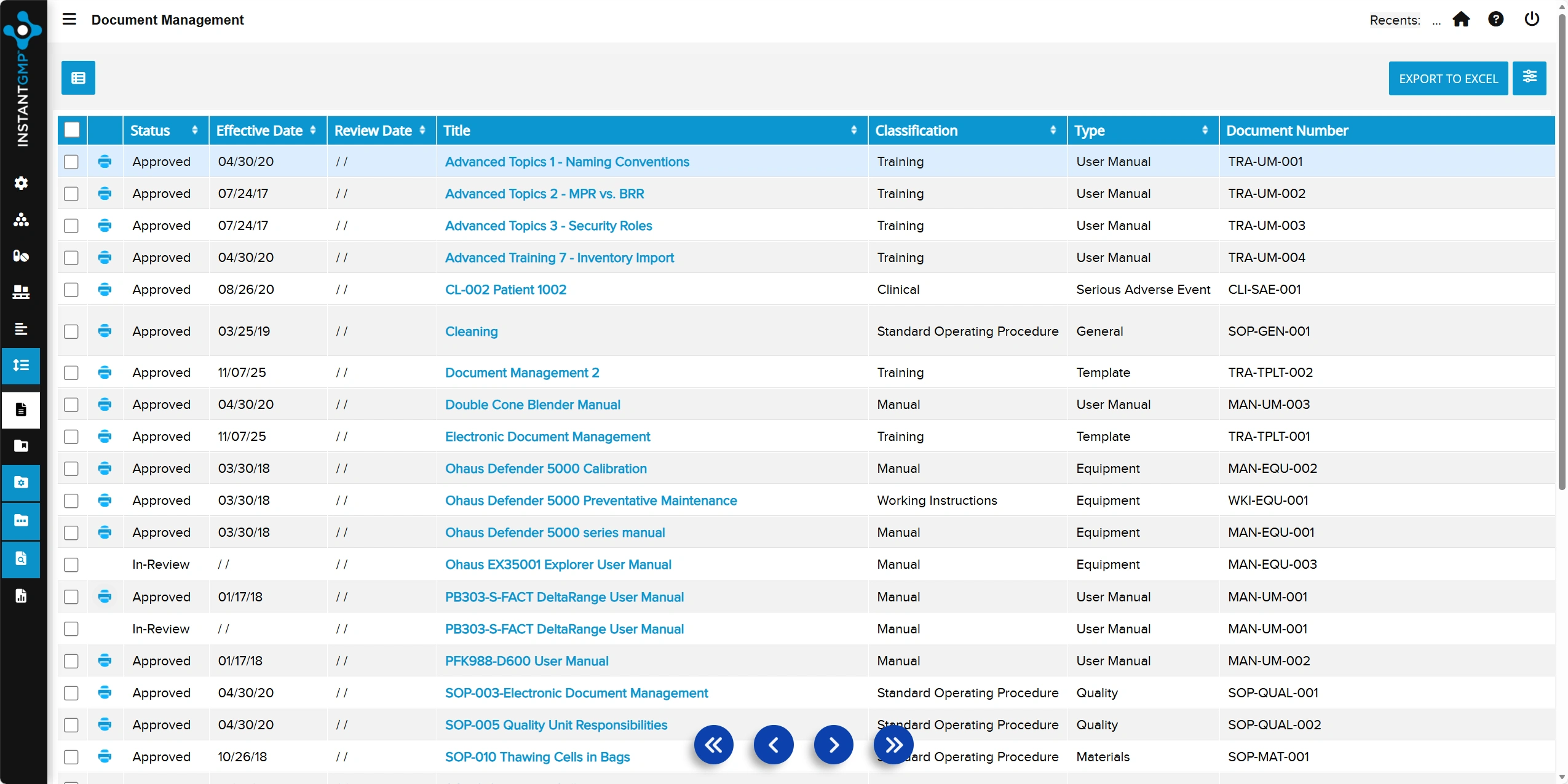
Centralized, audit-ready record of deviations with clear documentation of root cause, impact, and corrective actions
Streamlined tracking and closure of deviations to ensure compliance, accountability, and continuous improvement
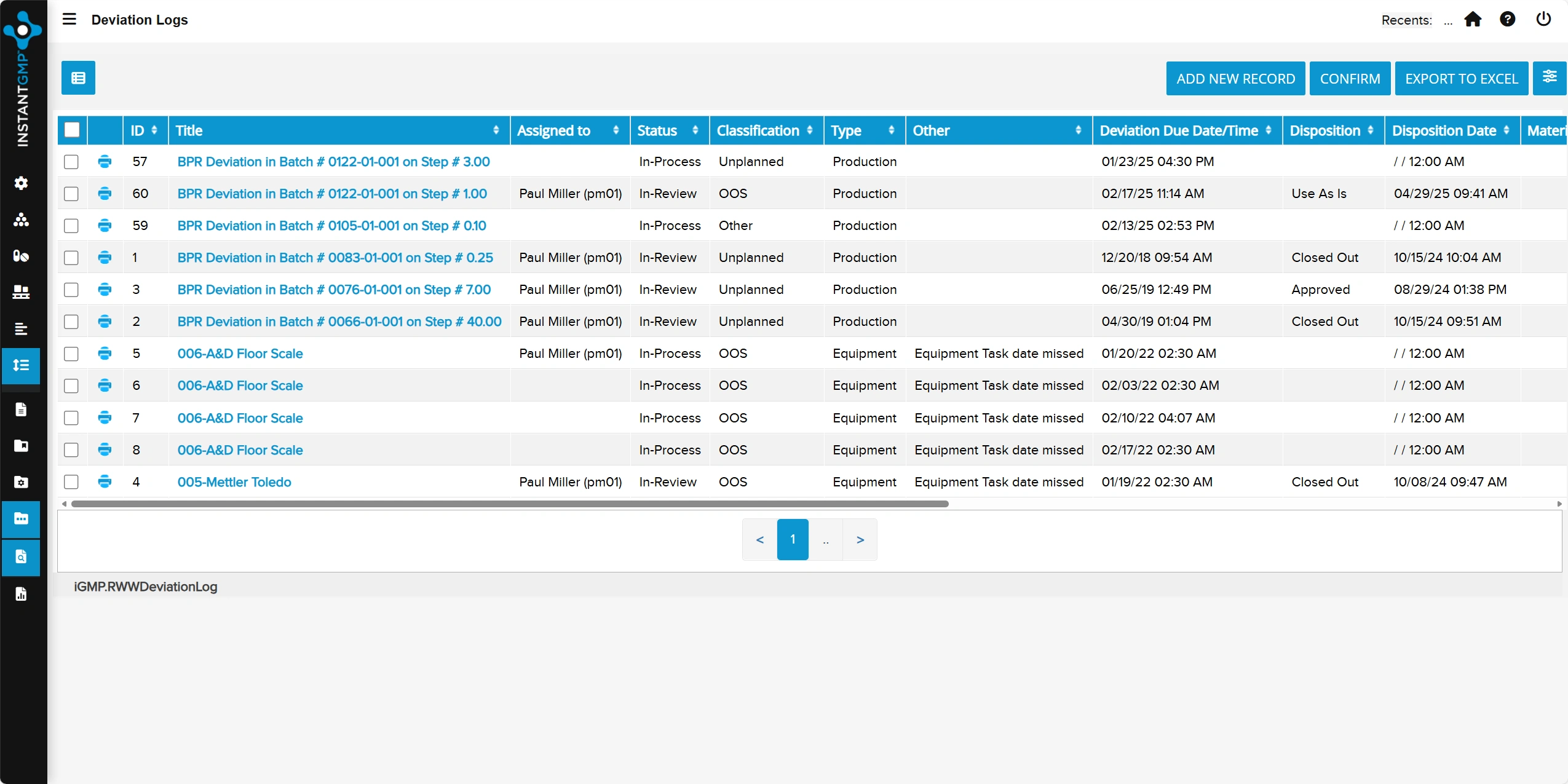
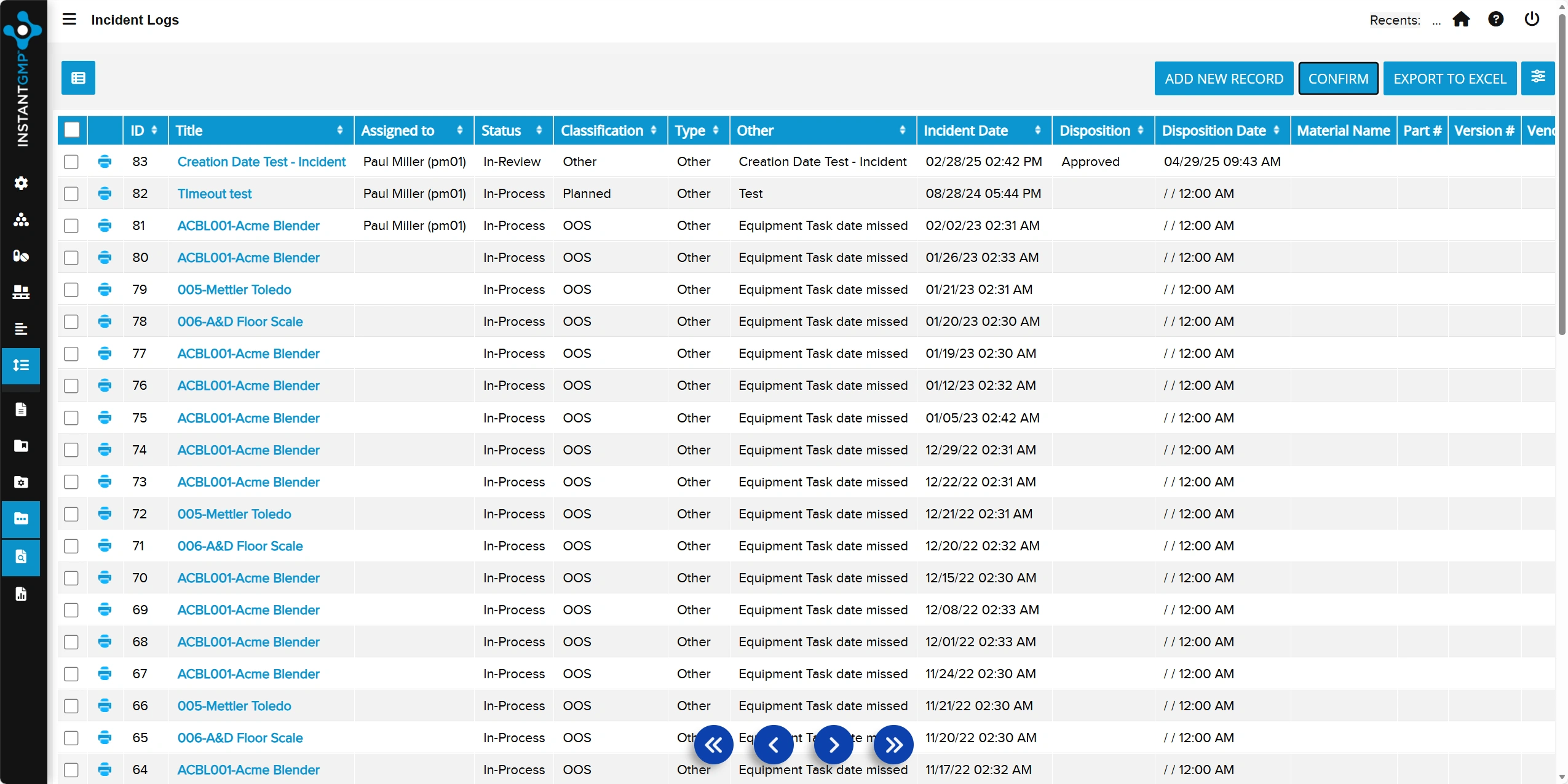
Centralized log for recording, investigating, and documenting incidents that impact quality or compliance
End-to-end tracking of actions and resolutions to prevent recurrence and support audit readiness
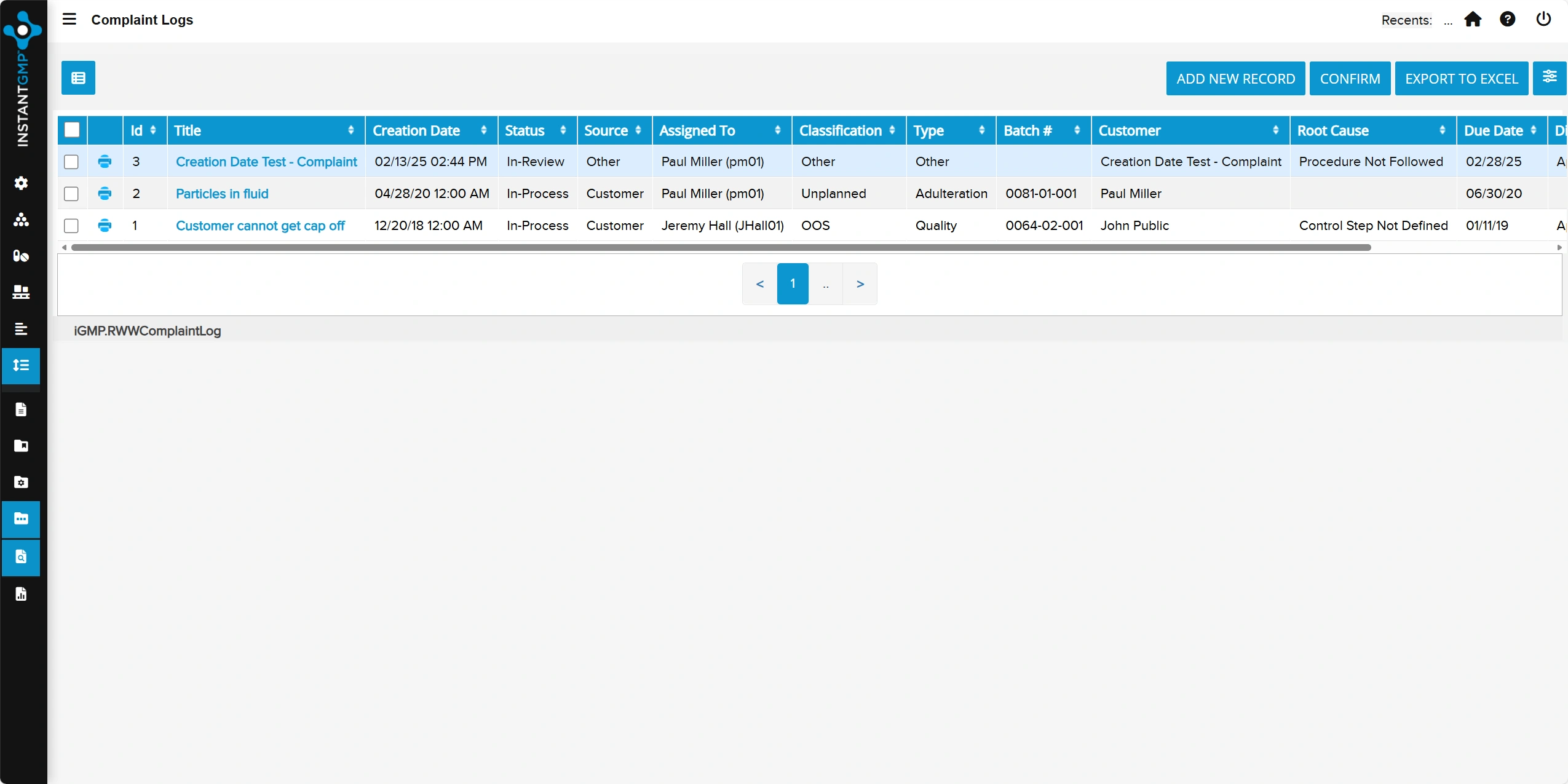
Centralized capture and investigation of customer complaints with documented outcomes and corrective actions
Traceable, audit-ready records that support regulatory compliance and continuous product improvement
Centralized tracking of corrective and preventive actions from identification through verification of effectiveness
Clear linkage to deviations, incidents, and complaints to drive compliance and continuous improvement
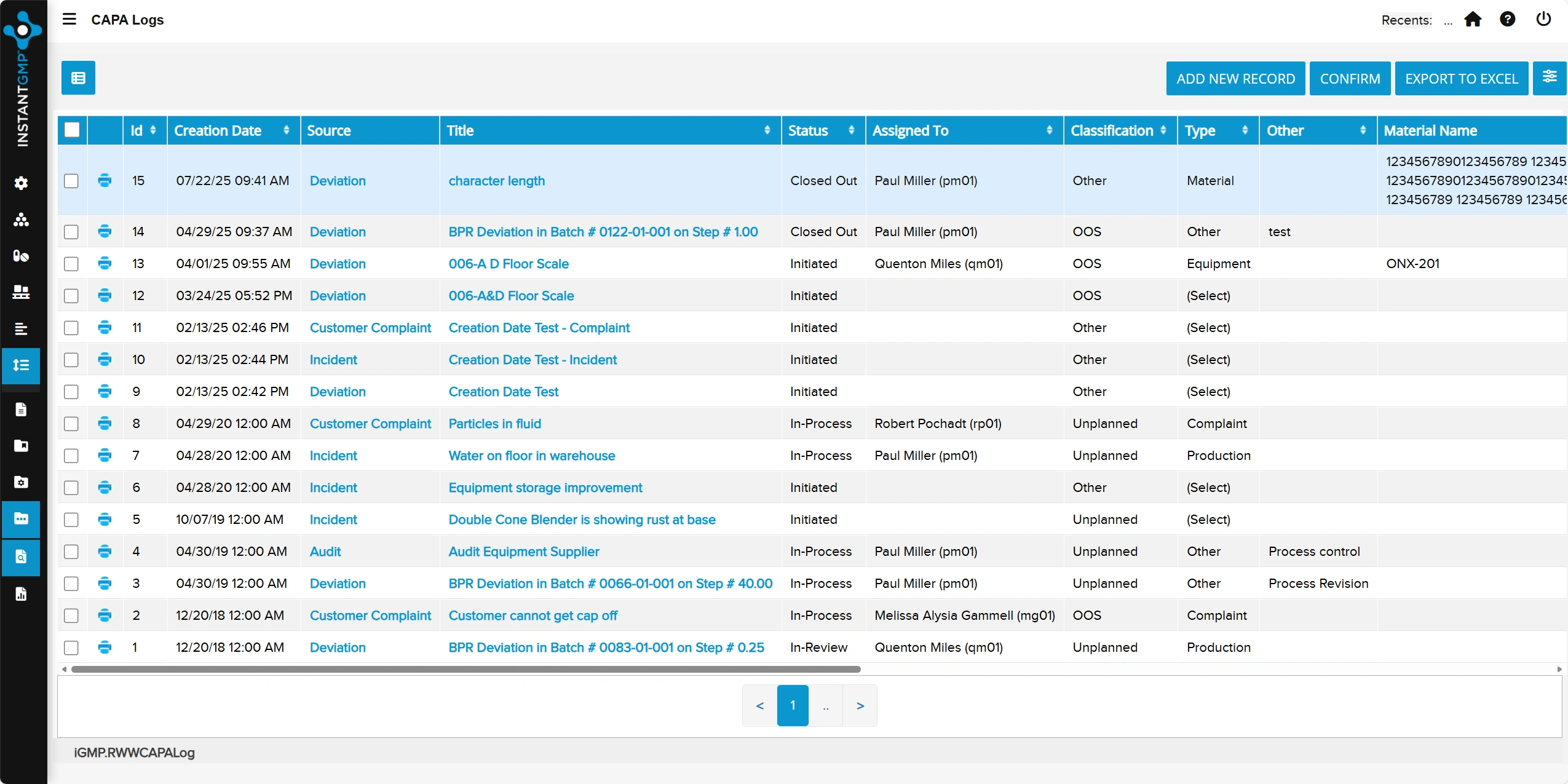
Structured documentation and approval of changes to processes, materials, equipment, and documents
Full traceability and impact assessment to ensure controlled, compliant change implementation
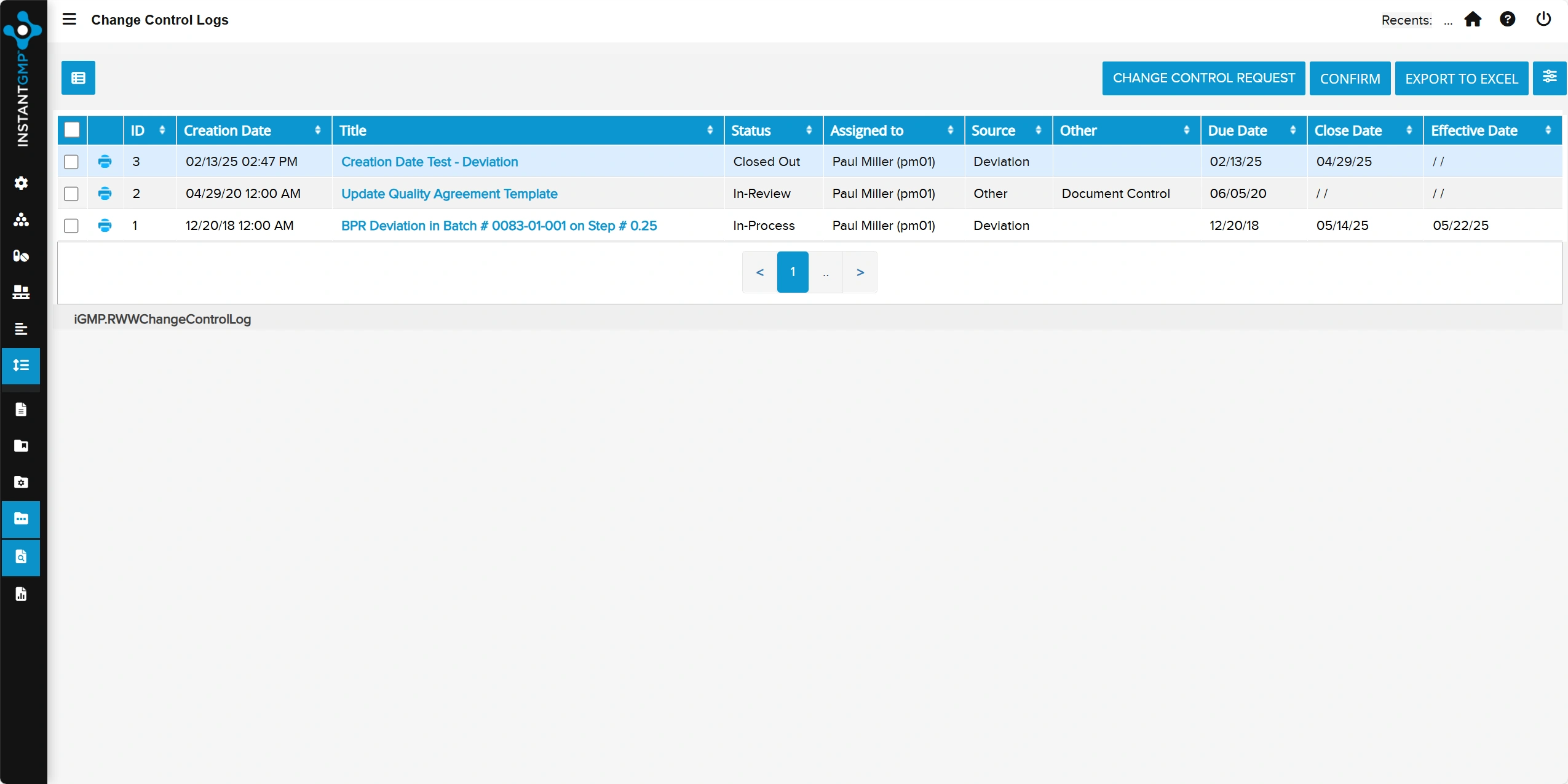
Complete, time-stamped record of user activity and system events for full transparency and traceability
Audit-ready evidence that supports data integrity, accountability, and regulatory compliance
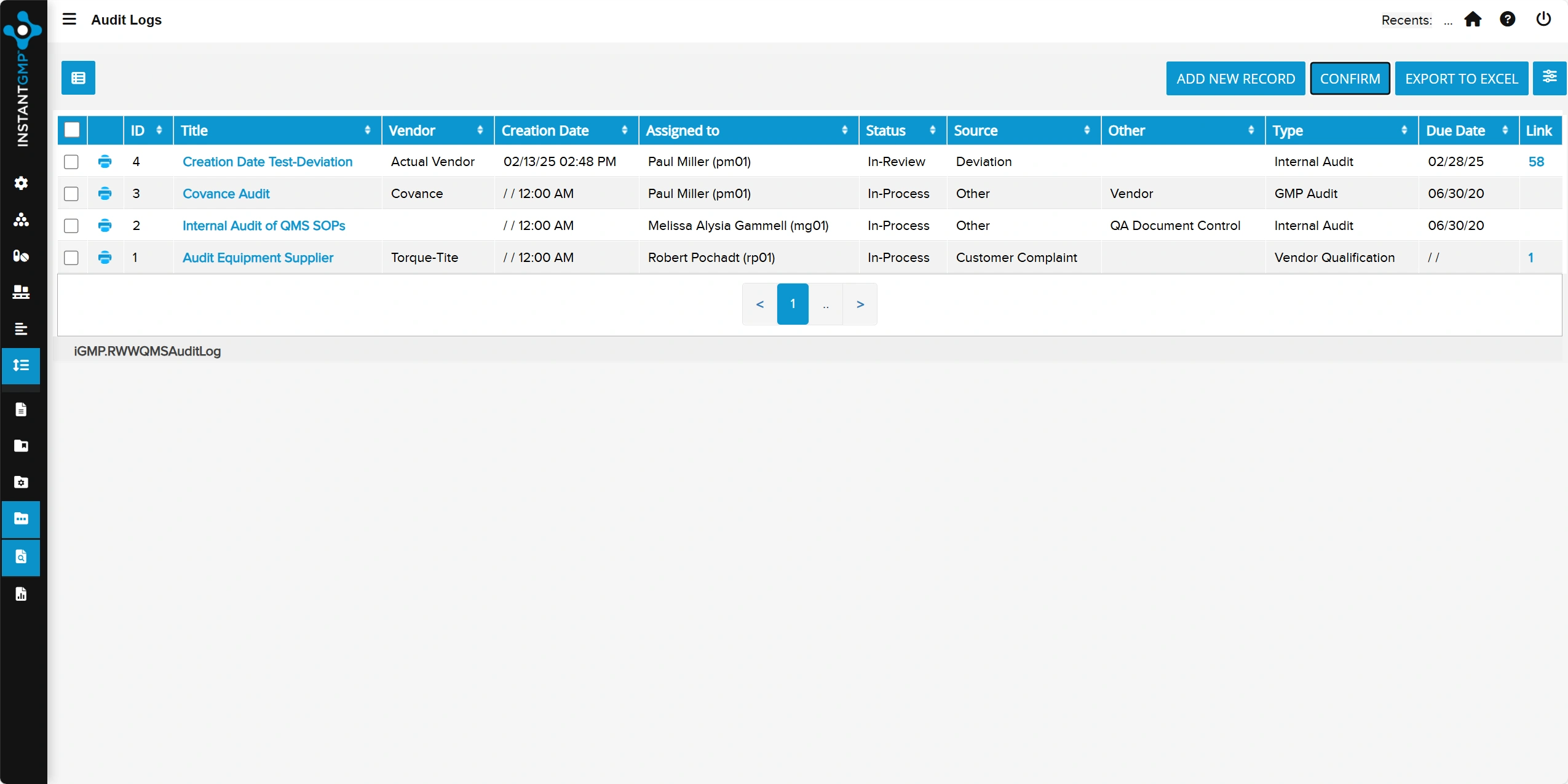
Centralized qualification and ongoing evaluation of vendors to ensure quality and compliance
Documented performance tracking, approvals, and re-evaluations to support audit readiness and supply chain control
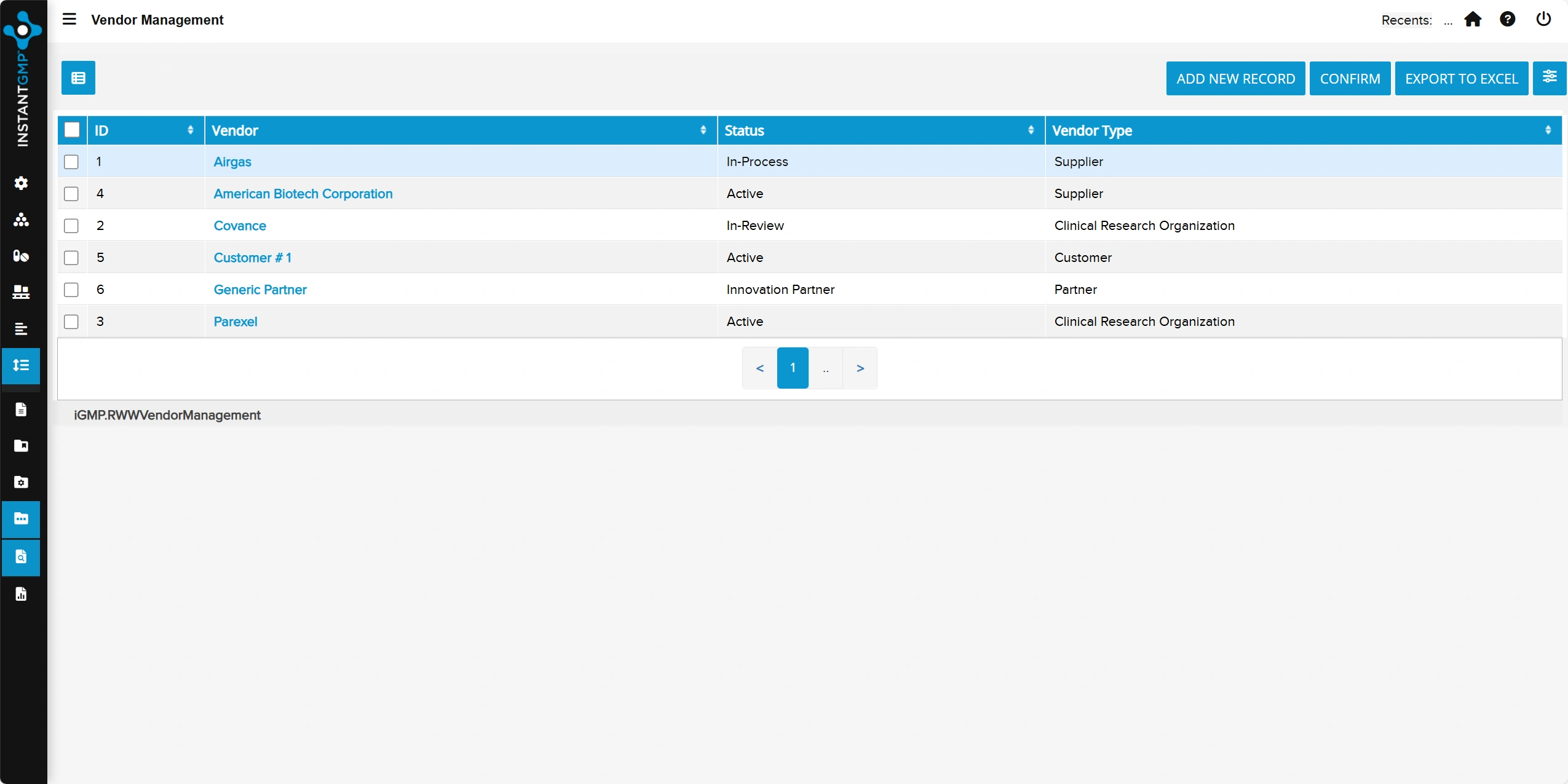
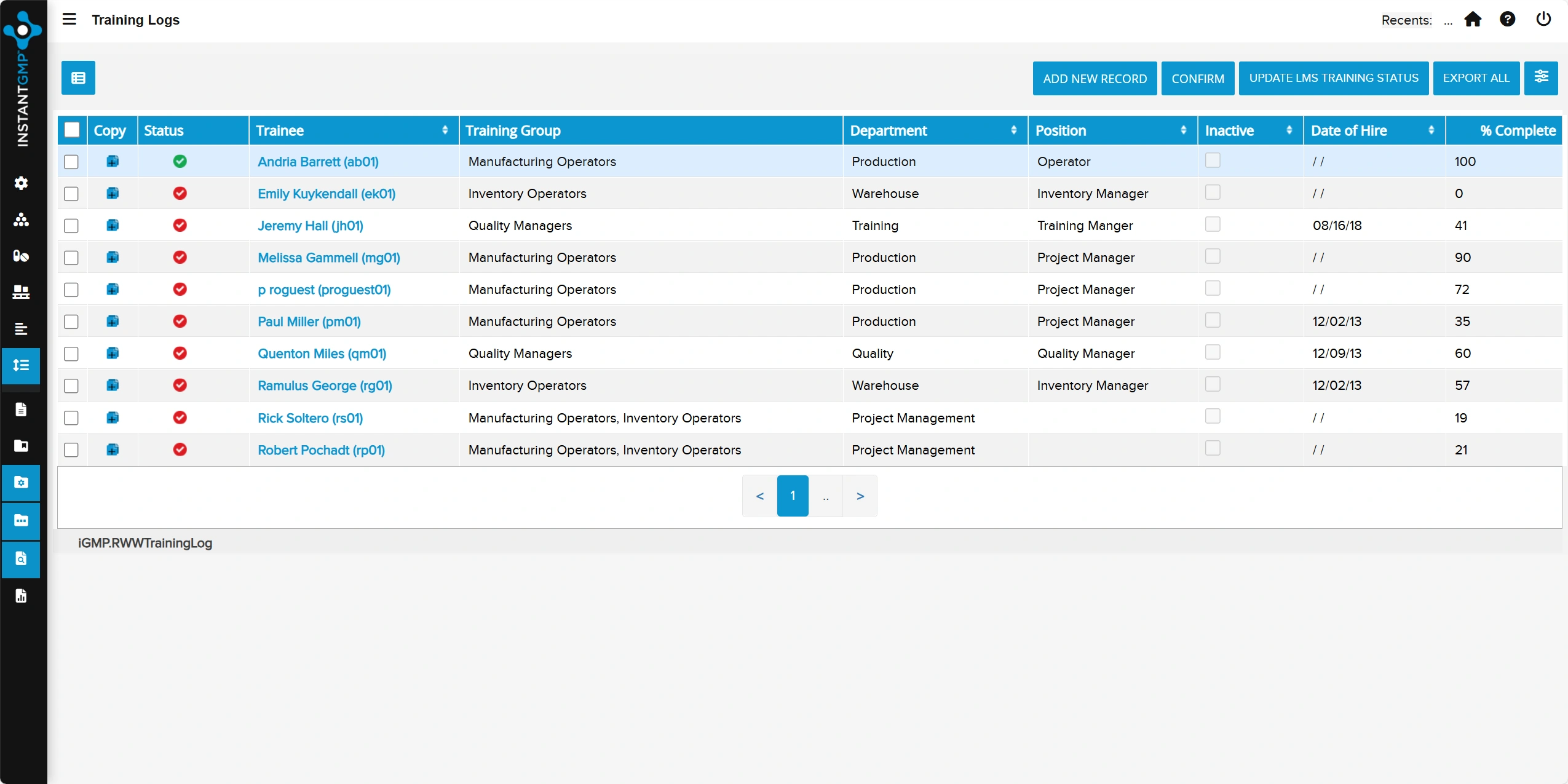
Centralized tracking of employee training, qualifications, and competency status by role
Audit-ready records with clear linkage to SOPs and required retraining to maintain compliance
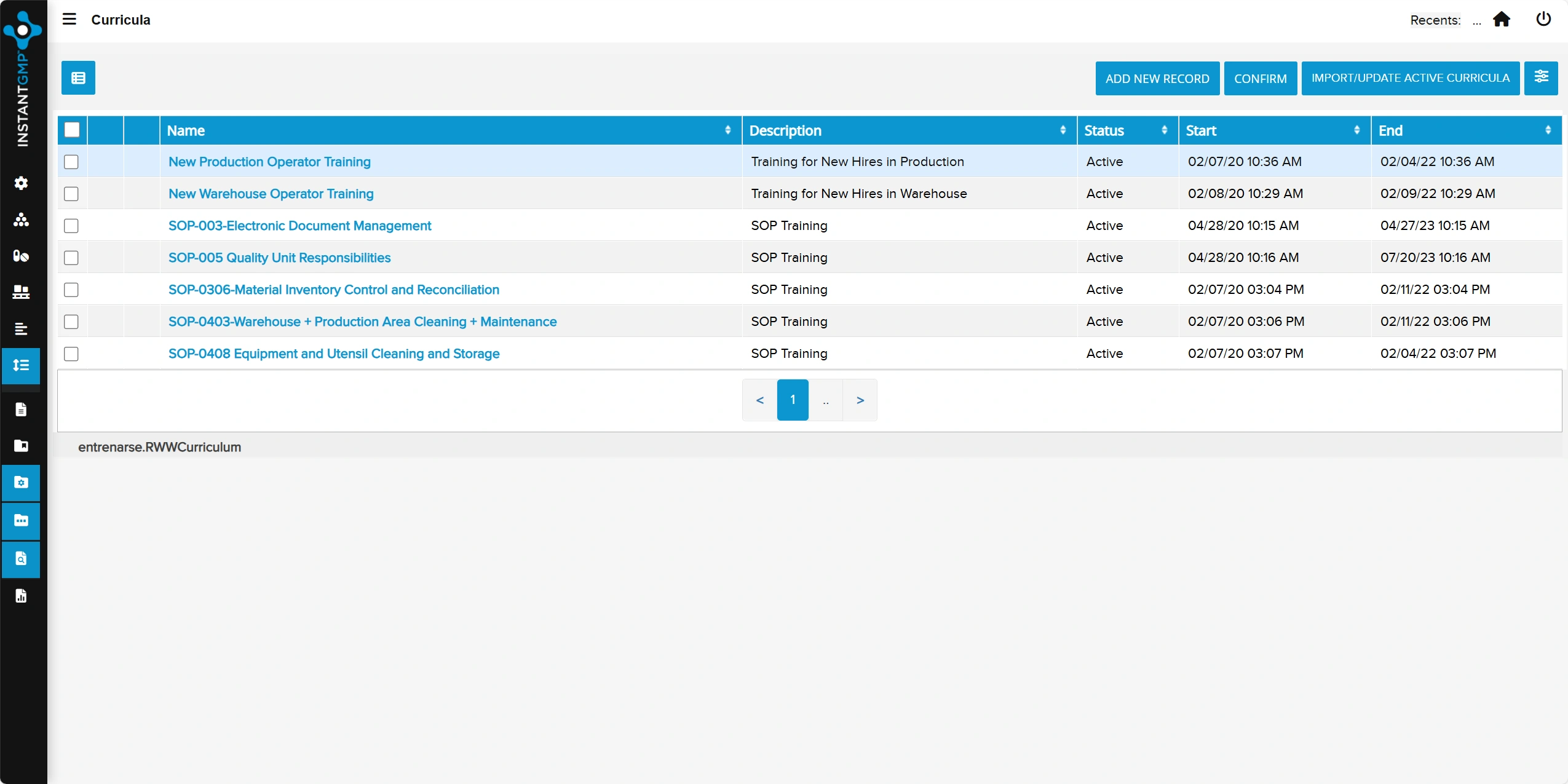
Centralized control of SOPs, records, and forms with versioning and electronic approvals
Secure access, full revision history, and audit-ready traceability to support compliance

Integrated learning management system to assign, deliver, and track GMP training by role
Automated reminders, completion tracking, and audit-ready training records to ensure compliance
A Quality Management System is the backbone that keeps a company’s operations organized, compliant, and repeatable. It brings all quality processes into one place, things like document control, training, audits, deviations, CAPAs, and change management, so teams can follow the right procedures every time and prove they did.
A QMS is essential because it turns quality from tribal knowledge and scattered paperwork into a controlled, traceable system. It reduces errors, prevents costly recalls or compliance issues, and makes inspections far less stressful. More than that, it creates consistency and confidence, helping companies scale faster, protect customers, and meet regulatory expectations without chaos piling up behind the scenes
InstantQMS adapts to your workflow, regardless of your product or process.
Ingredient tracking, safety documentation, batch consistency.
Supplier tracking, defect logging, continuous improvement cycles.
Component verification, process documentation, training control.
SOP standardization, safety procedures, audit readiness.
Product specification management, quality checks, labeling accuracy.
Change control, corrective actions, compliance documentation.
Manage all quality documents, records, and data live and in one secure system.
Ensure data integrity, complete traceability, and consistent workflows across your entire organization.
Strengthen operational consistency and readiness.
Get Simple, Trusted Compliance with InstantQMS to streamline processes, reduce errors, and ensure consistent product quality.